Built on science.
Engineered for outcomes.
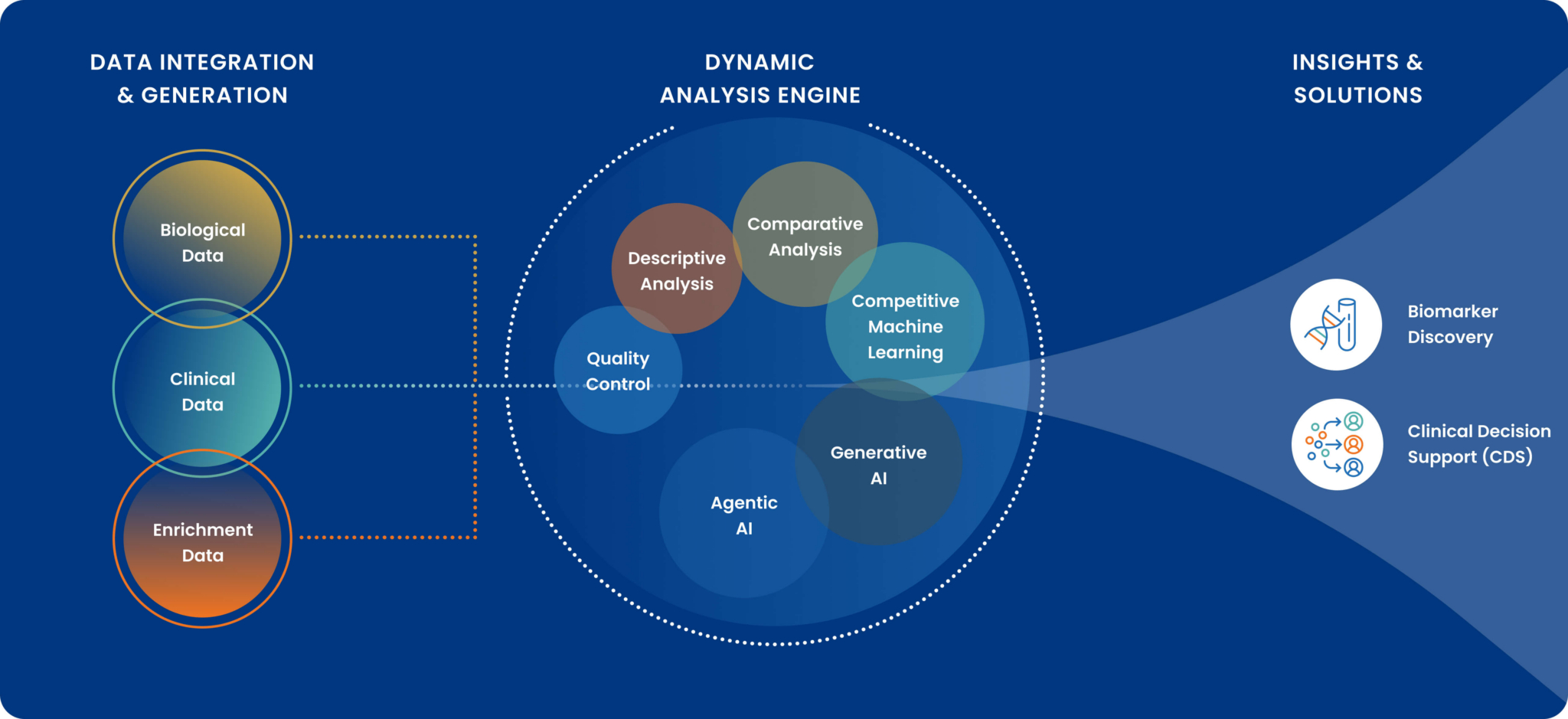
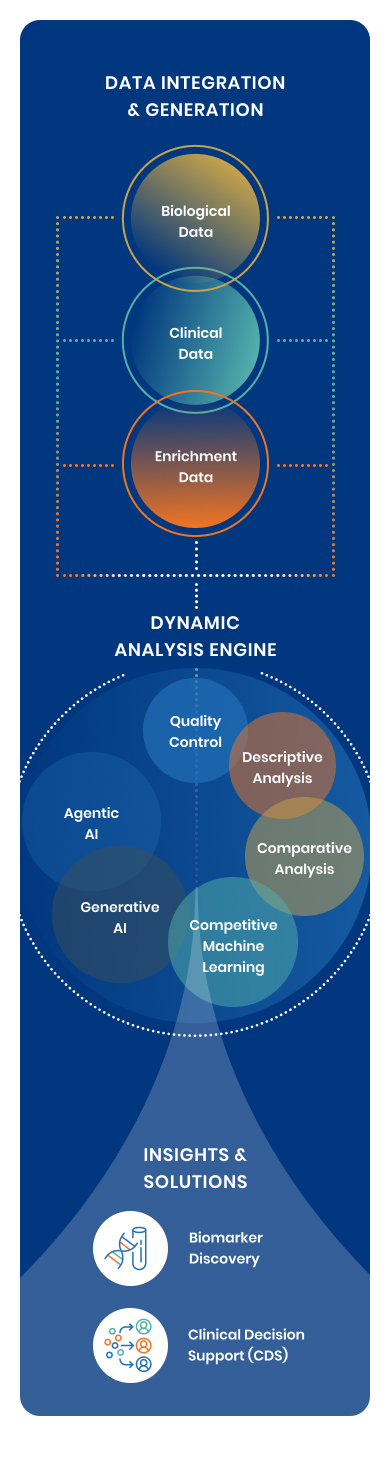
Decode combines an automated R&D engine with deep multiomics and clinical data expertise so your program moves from question to validated findings ready for your commercial roadmap.
Decode’s structured program model combines repeatable platform methodology with deep scientific expertise, producing results that hold up under commercial and regulatory scrutiny. Self-service tools require you to supply the science. Traditional engagements don’t compound. Decode solves both.
How the Platform Works
Every program runs through three structured phases with a defined scope, a fixed deliverable, and a clear decision gate at each step.
Every program runs through three structured phases with a defined scope, a fixed deliverable, and a clear decision gate at each step. Decode combines three things most R&D programs have to build separately: access to real patient data, an automated analytical engine, and deep scientific expertise in multiomics, clinical data, and health economics.
Programs start with a defined scope so partners can evaluate on real results before expanding. As programs demonstrate value, commercial structures naturally broaden into licensing, milestone-based arrangements, or revenue-sharing models. Partners often find that the first program opens the door to the next.
The platform improves with each program. Every engagement adds to a more calibrated pipeline, a richer proprietary data estate, and a deeper body of validated methodology. These are compounding assets that software alone cannot replicate. Your second program runs faster than your first.
As individual components mature, more of the pipeline runs autonomously, freeing the scientific team to focus on study design, biological interpretation, and commercial strategy.
01 Discovery Sprint
Define the question, map your data, agree on what success looks like before any platform work begins.
Inside Phase 1
02 Platform Development
Dozens of standardized analytical steps from ingestion through results, spanning automated processing and expert-configured analysis.
Inside Phase 2
03 Results For Your Next Stage
Validated findings structured for commercialization, regulatory review, investor diligence, or the next phase of your program.
Inside Phase 3
The Data Advantage
Proprietary Cohorts
IRB-approved, annotated patient cohorts across neurology and select disease areas, with established pathways to extend into new conditions. For the right programs, meaningful analysis can begin immediately.
Public & Licensed Data Acceleration
The platform is designed to work with publicly available multiomics datasets (DNA, RNA, protein) and licensed claims and EHR data, dramatically reducing the time and cost of de novo data collection.
Data Partnerships
When neither proprietary nor public data exists in sufficient depth, Decode’s established relationships with clinical networks, biorepositories, and sample collection partners provide a solution. The absence of data is a solvable problem, not a program blocker.
The Scientific Record
Pillar 1: Molecular Biomarker Discovery
94% Accuracy Distinguishing NMO from RRMS using whole-blood RNA sequencing. Journal of Molecular Diagnostics 2024Accuracy
AUC
Whole Blood
Pillar 2: Clinical Decision Support
76% spend capture vs. 43.5% and 36.5% with conventional methods. MS high-cost prediction. Communications Medicine 2025Decode
4-Month Historical
Prior Month
Federal Validation

Published & Peer-Reviewed
Disease-Agnostic Platform
Validated across multiple therapeutic areas. The platform extends to new conditions without rebuilding from scratch.









